Transforming Clinical Trials: How CTMS Drives Efficiency & Compliance
A modern Clinical Trial Management System (CTMS) puts planning, execution, and oversight all in one place, giving sponsors and investigators the clarity and control they need to succeed.
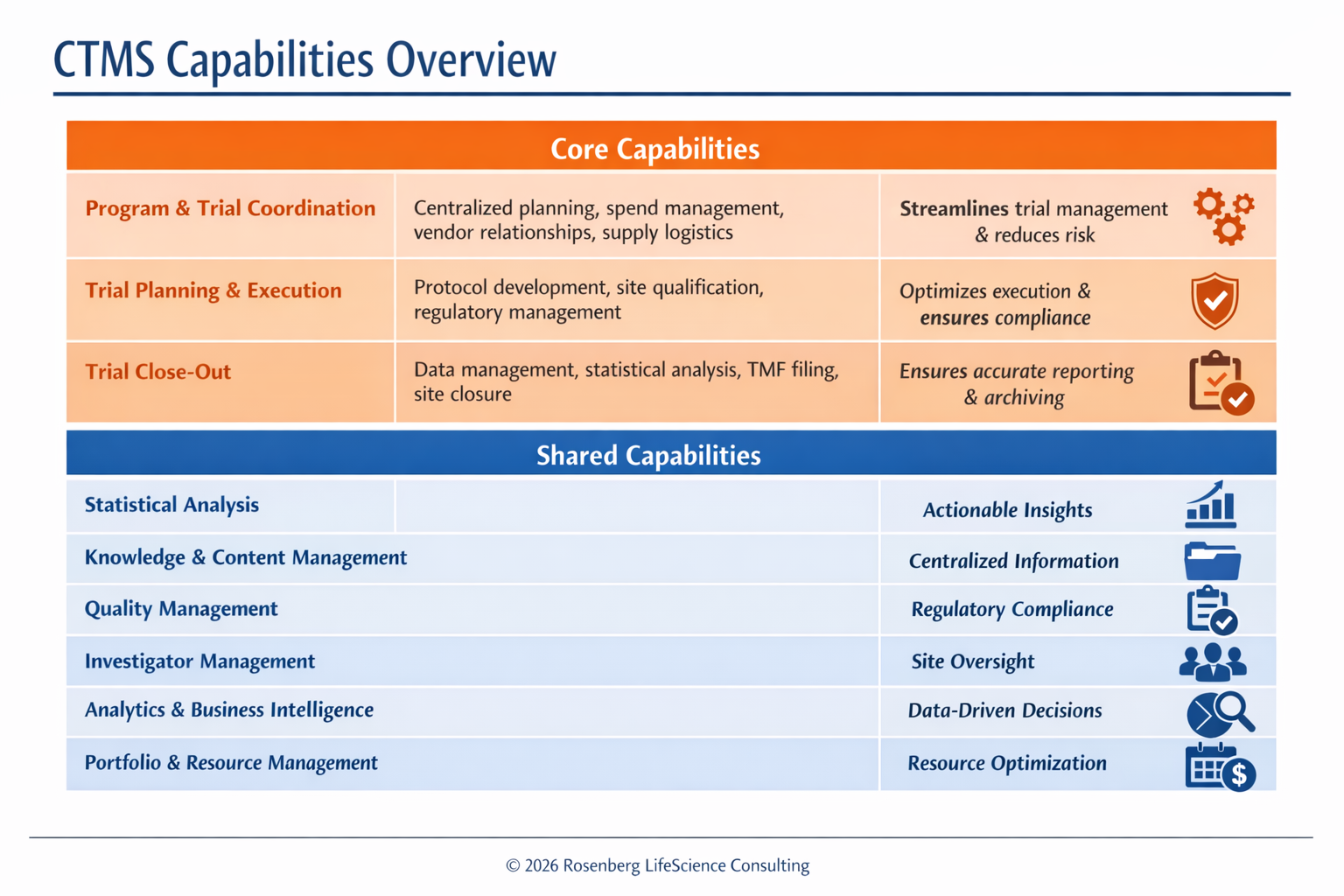
📊 Check out this snapshot of CTMS Core & Shared Capabilities [image attached] – from program coordination to portfolio management, it brings efficiency and transparency to every stage of the trial lifecycle.
CTMS Capabilities Overview
Why industry-leading CTMS platforms matter:
✅ Unify trial oversight, site coordination, and sponsor reporting in a single, secure interface
✅ Support regulatory readiness (FDA, EMA, ICH-GCP, GDPR) and integrate seamlessly with EDC, eTMF, and eSource
✅ Enable proactive compliance, automated deviation tracking, and audit-ready systems
✅ Deliver operational transparency and scalability—essential for patient safety and trial success
Core functional areas include:
📝 Trial planning & setup
🏥 Site & investigator management
👥 Participant monitoring & retention
📅 Scheduling & workflow automation
📂 Regulatory compliance & document management
💰 Financial tracking & budgeting
📈 Real-time monitoring & reporting
Best practices for successful implementation:
🎯 Treat CTMS as a strategic transformation, not just an IT project
🤝 Engage cross-functional teams: clinical ops, data, finance, regulatory, IT
📊 Define clear objectives and KPIs upfront
🔄 Commit to continuous improvement to stay compliant and efficient
Bottom line:
A modern CTMS isn’t just a tool—it’s a strategic enabler. By integrating planning, execution, monitoring, and close-out activities into a single platform, sponsors and investigators gain the insight and control needed to run trials efficiently, compliantly, and successfully.